Vitamin Labelling Requirements on UK Food Labels: A Complete Guide
Vitamins sell products. “Source of vitamin D,” “high in vitamin C,” “contains B12” – these phrases connect with health-conscious consumers and can be a legitimate competitive advantage. But vitamin labelling on food labels is tightly regulated. Use the wrong threshold, wrong units, or wrong wording, and you’re non-compliant before the product leaves the factory. This guide explains what UK food businesses need to know about labelling vitamins correctly and legally.
Why Vitamin Labelling Rules Matter
Vitamin labelling appears on food labels in two distinct contexts:
- Mandatory declaration – when vitamins are added to a food, or when a nutrition or health claim is made about a vitamin, it must appear in the nutritional information table.
- Voluntary claim – when a business wants to highlight that a product is a “source of” or “high in” a particular vitamin, specific thresholds must be met and specific wording used.
Get either wrong and you risk enforcement action from Trading Standards. Since October 2024, the first step is typically an improvement notice rather than immediate prosecution – but repeated non-compliance or deliberate violations can still result in criminal proceedings.
Which Vitamins Can Appear on UK Food Labels?
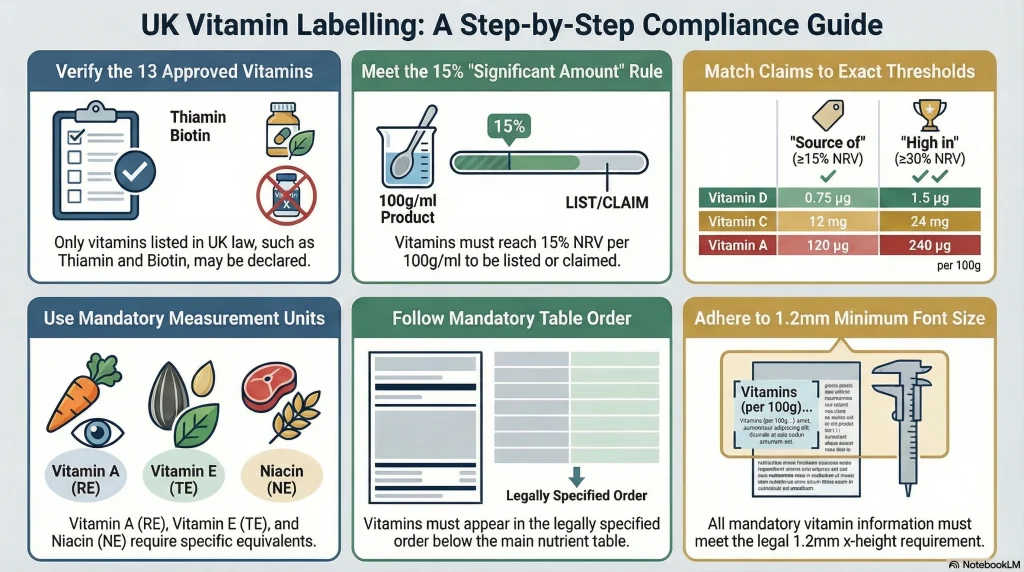
UK food law (retained EU Regulation 1169/2011 on Food Information to Consumers) specifies 13 vitamins that may be declared in the nutrition table, along with their legal names and Nutrient Reference Values (NRVs).
The 13 Vitamins Recognised for UK Food Labelling
These are the vitamins recognised for vitamin labelling purposes and their NRVs:
| Vitamin | Legal Name (for labels) | NRV |
|---|---|---|
| Vitamin A | Vitamin A | 800 µg |
| Vitamin D | Vitamin D | 5 µg |
| Vitamin E | Vitamin E | 12 mg |
| Vitamin K | Vitamin K | 75 µg |
| Vitamin C | Vitamin C | 80 mg |
| Thiamin (B1) | Thiamin | 1.1 mg |
| Riboflavin (B2) | Riboflavin | 1.4 mg |
| Niacin (B3) | Niacin | 16 mg |
| Vitamin B6 | Vitamin B6 | 1.4 mg |
| Folic Acid (B9) | Folic Acid | 200 µg |
| Vitamin B12 | Vitamin B12 | 2.5 µg |
| Biotin (B7) | Biotin | 50 µg |
| Pantothenic Acid (B5) | Pantothenic acid | 6 mg |
Only vitamins from this list may be declared in the nutrition information table. Other substances sometimes marketed as vitamins – such as choline or inositol – do not appear on this list and cannot be declared as vitamins on a standard food label.
Units Matter in Vitamin Labelling
Specific vitamins must be expressed in particular units:
- Vitamin A must be expressed as micrograms of retinol equivalents (RE)
- Vitamin E must be expressed as milligrams of d-alpha-tocopherol equivalents (TE)
- Niacin (B3) must be expressed as milligrams of niacin equivalents (NE)
Using the wrong unit is a labelling error even if the quantity itself is correct.
The “Significant Amounts” Rule
A vitamin can only be declared in the nutrition table, or used in a claim, if it is present in a significant amount. The definition depends on context.
For foods generally: a vitamin must be present at a level of at least 15% of the NRV per 100g (for solids) or 15% NRV per 100ml (for liquids) to qualify as a “significant amount.”
For single-serve portion products: the 15% NRV threshold applies to the amount in the portion that would reasonably be consumed, not the per-100g figure.
If a vitamin is present below this threshold, you cannot list it in the nutrition table or make any claim about it – even in passing. An ingredient list can reference the vitamin’s presence, but it cannot be promoted.
“Source of” vs “High in”: Vitamin Claim Thresholds
If you want to feature a vitamin prominently on pack – on the front label, in a strapline, or as a selling point – you must use the authorised claim wording from the GB NHC Register and your product must meet the corresponding threshold:
| Claim | Threshold |
|---|---|
| “Source of [vitamin]” | ≥15% of NRV per 100g (solids) or per 100ml (liquids) |
| “High in [vitamin]” | ≥30% of NRV per 100g (solids) or per 100ml (liquids) |
Some practical examples:
- To claim “source of vitamin D,” your product needs at least 0.75 µg of vitamin D per 100g (15% of 5 µg NRV)
- To claim “high in vitamin D,” it needs at least 1.5 µg per 100g (30% of 5 µg NRV)
- To claim “source of vitamin C,” you need at least 12 mg per 100g (15% of 80 mg NRV)
- To claim “high in vitamin C,” you need at least 24 mg per 100g (30% of 80 mg NRV)
The claim wording must match the authorised text on the GB NHC Register exactly. Creative alternatives like “packed with vitamin C” or “vitamin C rich” are not authorised claim wording and risk non-compliance.
Declaring Vitamins in the Nutrition Table
When a vitamin must be included in the nutrition table (because it is added to the food or a claim is being made), the declaration must meet these requirements:
- Expressed per 100g or per 100ml as standard
- Can additionally be expressed per portion alongside the per-100g/ml figures
- Must include the % of NRV (Reference Intake) – the label must state: “Reference intake of an average adult (8400 kJ / 2000 kcal)”
- Vitamins appear after the main mandatory nutrients as supplementary information within or below the nutrition table
- They must appear in the order specified in law – you cannot rearrange them for design reasons
The term to use on labels is “reference intakes” (formerly guideline daily amounts or recommended daily allowances). Using “NRV” is not prohibited, but “reference intakes” is the preferred regulatory term.
Vitamin Labelling for Food Supplements
Food supplements containing vitamins are subject to their own specific rules, governed by the Food Supplements (England) Regulations 2003 and equivalent devolved legislation, as well as the general food information rules. Vitamin labelling requirements for supplements are more prescriptive than for standard foods.
Mandatory Information on Food Supplement Labels
A compliant food supplement label must include:
- The legal name “Food Supplement” – this is mandatory and must appear on the label
- A list of ingredients in descending order of weight, using full chemical names for vitamins (e.g. “cholecalciferol” for Vitamin D3, “ascorbic acid” for Vitamin C)
- The amount of each vitamin provided per recommended daily intake (dose), in the units specified by law
- The % NRV for each vitamin present at ≥15% NRV per recommended daily intake
- The recommended daily intake and a warning not to exceed it
- A statement that supplements should not replace a varied diet
- A notice to keep the product out of the reach of young children
- Best before date, batch number, storage conditions, net weight, and business operator details
There is no requirement to register food supplements with any UK authority before sale. However, it is entirely the responsibility of the manufacturer, importer, or retailer to ensure compliance.
Vitamin Labelling Units for Supplement Products
The same unit rules that apply to food labels apply to supplements – but with additional precision required. For supplements, the amount per recommended daily intake must be declared in the units specified in law, not in alternative units of convenience. Vitamin D3 declared in IU rather than µg, for example, is a compliance failure even if the amount is equivalent.
Why Accurate Vitamin Labelling Protects Your Business
Non-compliant vitamin labelling is one of the most common reasons food supplement businesses receive improvement notices from Trading Standards. Getting units, thresholds, and claim wording right protects you from enforcement action, but it also builds consumer trust – particularly in a supplement market where shoppers are increasingly scrutinising labels carefully.
Note: the word “Organic” should not appear anywhere on a food supplement label unless the product holds certified organic status.
Health Claims Linked to Vitamins
Many of the most commonly used authorised health claims in the UK relate to vitamins. These are drawn from the GB NHC Register and must be used with exact authorised wording. Some examples:
| Vitamin | Example Authorised Health Claim |
|---|---|
| Vitamin D | “Vitamin D contributes to the normal function of the immune system” |
| Vitamin C | “Vitamin C contributes to the reduction of tiredness and fatigue” |
| Vitamin C | “Vitamin C contributes to normal collagen formation for the normal function of skin” |
| Folic Acid | “Folate contributes to maternal tissue growth during pregnancy” |
| Vitamin B12 | “Vitamin B12 contributes to normal energy-yielding metabolism” |
| Vitamin E | “Vitamin E contributes to the protection of cells from oxidative stress” |
| Calcium + Vitamin D | “Calcium and vitamin D are needed for normal bone structure” |
To make any of these health claims, the product must first meet the threshold conditions – usually that it qualifies as at least a “source of” the relevant vitamin (≥15% NRV). You cannot paraphrase these claims. “Helps your immune system” is not equivalent to “contributes to the normal function of the immune system” – the former is unapproved wording and potentially non-compliant.
EU/UK Divergence for Vitamin Claims
The framework for vitamin-related health claims was largely settled before Brexit, and the risk of divergence is lower for vitamin claims than for botanical ingredient claims, because the science for vitamins is well-established and the list of authorised claims is stable.
However, a few things to watch:
- New EU health claims approved after January 2021 are not automatically valid in GB
- Northern Ireland continues to use EU rules; businesses operating in both markets should maintain dual compliance
- UK-EU SPS re-alignment (mid-2027) is expected to converge the two registers again. For vitamin-specific claims, this is unlikely to require significant label changes – but it should be confirmed once implementation guidance is published
Common Vitamin Labelling Mistakes
Using the wrong units. Declaring vitamin A in milligrams rather than micrograms of retinol equivalents, or failing to express vitamin E as tocopherol equivalents, is a technical non-compliance even if the number is approximately right.
Listing below-threshold vitamins. If a vitamin is present at less than 15% NRV per 100g, it cannot appear in the nutrition table at all as a declared vitamin.
Making implied vitamin claims without meeting thresholds. Listing “enriched with vitamin B12” in a product description without the required level in the nutrition information is an implied claim without substantiation.
Confusing NRV with RDA or GDA. These older terms are no longer correct on food labels. Use “reference intakes” (RI) or “nutrient reference values” (NRV). Using “RDA” on a label is outdated and could attract enforcement attention.
Front-of-pack claims not matched on the back. If your front-of-pack highlights “source of vitamin D,” the nutrition table on the back must include the vitamin D declaration confirming it meets the threshold. Inconsistency between the front and back of pack is a recognised compliance failure.
Regulatory Sources
- GB NHC Register: gov.uk/government/publications/great-britain-nutrition-and-health-claims-nhc-register
- FSA Nutrition Labelling Guidance: food.gov.uk/business-guidance/nutrition-labelling
- FSA Food Supplements Guidance: food.gov.uk/business-guidance/food-supplements
- DHSC Nutrition Legislation Information Sheet: gov.uk/government/publications/nutrition-legislation-information-sources
- Food Supplements (England) Regulations 2003: legislation.gov.uk/uksi/2003/1387
- Retained FIC Regulation (EU) 1169/2011: legislation.gov.uk/eur/2011/1169
- NRV Table and Technical Guidance: gov.uk/government/publications/technical-guidance-on-nutrition-labelling
Frequently Asked Questions
Do I have to list vitamins in the nutrition table?
Only if vitamins are added to your food, or if you are making a nutrition or health claim about a vitamin. If vitamins occur naturally and you are making no claim about them, declaration is voluntary – but once you declare them, the full vitamin labelling rules apply.
Can I say “contains vitamin D” without meeting the 15% NRV threshold?
“Contains vitamin D” is an implied nutrition claim that suggests a nutritional benefit. It should only be used where the vitamin D content meets the threshold conditions. If you’re simply listing it as an ingredient that’s different – but using it as a selling point without meeting the threshold is a compliance risk.
My supplement has very high vitamin levels – is that a problem?
UK law does not currently set maximum vitamin levels in food supplements. However, the Expert Group on Vitamins and Minerals (EVM) published safe upper levels as guidance, and products significantly exceeding these may attract regulatory scrutiny. Always include “do not exceed recommended dose” warnings.
I’m fortifying my product with vitamin D. What do I need to do?
Fortification is covered by retained Regulation (EC) 1925/2006 on the addition of vitamins and minerals to foods. You must use approved vitamin forms, must not add vitamins to alcohol, and must declare the added vitamin in the nutrition table. A notification process may apply in some cases – check current FSA guidance.
Can I use “vitamin complex” or similar grouping terms on my label?
No. Each vitamin must be declared individually by its legal name. You cannot group them under a collective term in the nutrition table or in the ingredients list – full chemical names for vitamins are required in food supplement ingredients lists.
What font size do I need for vitamin information on the label?
Mandatory label information – including the nutrition table and required vitamin declarations – must use a minimum font size of 1.2mm for lower-case letters (x-height). On small packs, this is a real design constraint that needs to be factored in from the outset.
Does the NRV change for children’s products?
The NRVs used in standard food labelling are based on an average adult. For products specifically marketed for children, different reference values may apply under the legislation for foods for specific groups (retained Regulation EU 609/2013). Take specialist advice if producing products specifically for children.
How Positive ID Labels Can Help
Vitamin labelling compliance is, in large part, a print and design problem. You can have the nutrition science absolutely right – correct thresholds, correct wording, correct NRV percentages – and still fail at the label stage if the text is too small, the nutrition table is poorly laid out, or the front-of-pack claim doesn’t align with the back.
This is where 20+ years of food label printing experience makes a difference.
Our Xeikon digital press (1200dpi) handles the precision small-text printing that vitamin-dense labels demand. Nutrition tables with multiple vitamins, percentage reference intakes, footnote text, and front-of-pack claim wording all need to be legible at the legally required minimum 1.2mm x-height – on small packs for food supplements or fortified products, that’s a tight brief. The Xeikon handles it cleanly on premium papers and materials.
For food supplement bottles and jars, our Screen UV inkjet press produces durable, waterproof polypropylene labels that remain legible and non-peeling in the humid conditions of a bathroom cabinet or kitchen shelf.
If you’re launching a new fortified product or reformulating an existing one, our Jetrion laser die-cutting press lets you print short trial runs without committing to volume – ideal for testing label layouts before the full production run.
Most orders ship within 3 – 5 days. Call 01332 864895 or use our enquiry form for a free quote and sample discussion. We’ll help you get compliant vitamin labelling right first time.
We’ve had a great experience working with this supplier, especially with Natalie - she is always very responsive, professional, and friendly. The pricing is highly competitive and the sales service has been excellent from start to finish.If I could offer one suggestion, it would be to improve the account and invoice management process. It would be really helpful if there were a system for clients to easily track their past orders and invoices in one place.Overall, very pleased with the service and look forward to continuing our collaboration.
Absolutely brilliant service from start to finish. The team are brilliant and the product was excellent quality and exactly what I wanted. Natalie is amazing as are her colleagues. Will definitely be a returning customer. Thank you
Ive used Positive ID Labelling Ltd for all our egg box labels for many years now. Natalie is always above and beyond helpful and the service is exceptionally fast and efficient.
I inadvertently ordered the incorrect label guns which was quickly sorted by Natalie. She was excellent, customer service at its best! Thank you once again
All food labelling guides are provided in good faith for information purposes only and do not constitute legal advice. For specific compliance questions about specific labelling laws, contact a specialist or contact your local Trading Standards authority. Read our Regulatory Information Disclaimer
